Variant Reclassification
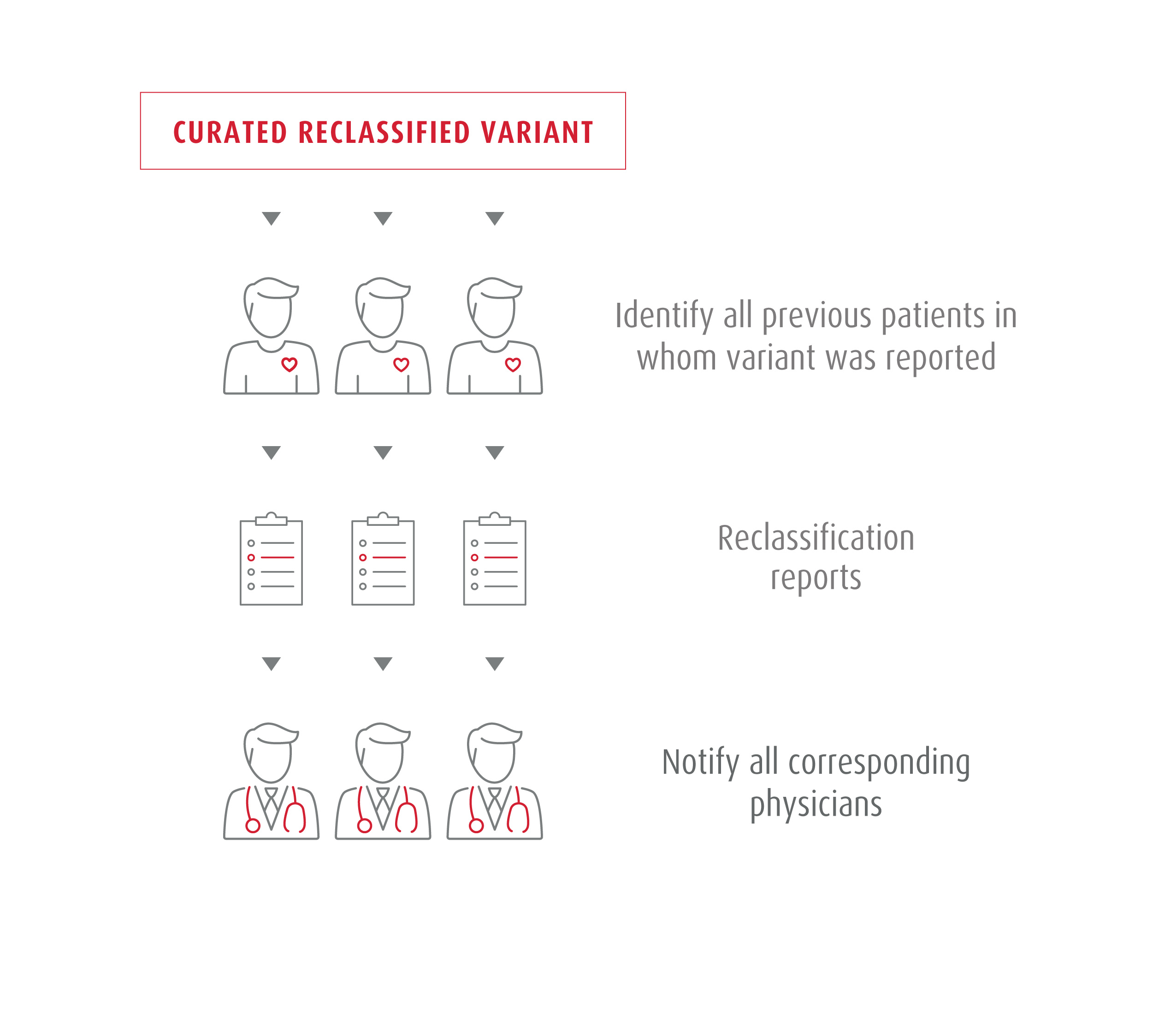
All diagnostic results from our tests are automatically entered into our variant reclassification program. This program supports the identification of new genetic evidence. We notify physicians automatically and free of charge if a new classification has an impact on a previous diagnosis.
Lifelong Commitment to Patient Safety Through Systematically Re-Evaluated Classifications
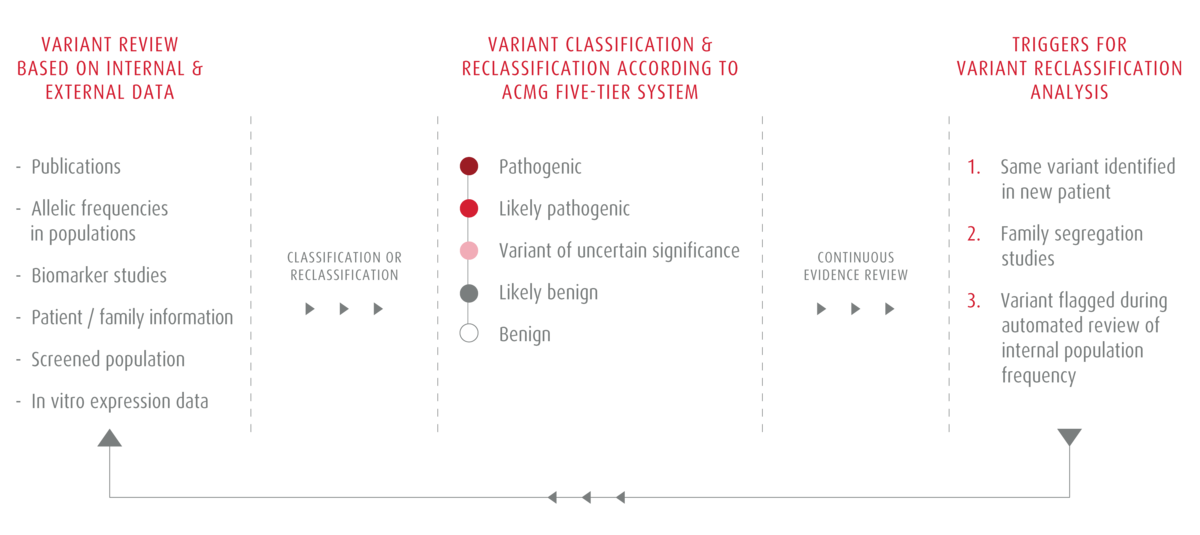
New disease-causing variants are identified every year. Consequently, classifying variants is an on ongoing process. At CENTOGENE, we have a dedicated team of experts who monitor medical literature and update our Bio/Databank accordingly. We use internationally recognized American College of Medical Genetics and Genomics (ACMG) guidelines to evaluate new evidence. We combine it with our patient data to make informed decisions when classifying newly identified genetic variants.
Once a variant is classified, every new observation, whether via internal or external research, is monitored and used to also re-evaluate past classification decisions systematically. New evidence can sometimes suggest a change in diagnosis or treatment. We communicate all re-classification decisions to our referring physicians, and any re-classification affecting a genetic diagnosis is shared as soon as possible.
Advantages of Our Variant Reclassification Program
- Proactive notification of every patient affected by the reclassification
- Based on a unique combination of broad patient data - genetic, biochemical, and clinical
- Variant data derived from a large and diverse cohort of patients
- Stringent data curation and validation process
Downloads & Resources

Reclassification Report
Carrier testing in the NSD1 (OMIM®: 606681) gene. Reclassified from uncertain clinical significance (class 3) to likely pathogenic (class 2).
Related Webinars
Niemann-Pick tipo C: Dos genes – muchos cuadros clínicos – un biomarcador unificador
Mira nuestro webinar sobre Niemann-Pick tipo C, un raro trastorno genético progresivo caracterizado por la incapacidad del cuerpo para transportar el colesterol y otras sustancias grasas (lípidos) […]
Niemann-Pick Type C: Two Genes - Many Clinical Pictures - One Unifying Biomarker
Watch our webinar “Niemann-Pick type C: Two Genes – Many Clinical Pictures – One Unifying Biomarker” hosted by Prof. Peter Bauer. Throughout the webinar, he will provide an overview of this rare […]
Gaucher Disease – Update on diagnosing and therapy monitoring
Knowing Gaucher Disease (GD), the most common lysosomal storage disorder, is crucial when being faced with unspecific abnormalities in many organs like brain, liver, spleen and bones. Right at […]
Get in Touch With Our Customer Support
Our consultation service is available in several languages.
+49 (0) 381 80 113 - 416
Mon. – Fri. 7 a.m. – 6:30 p.m. CET
Sat. 8 a.m. – 12 p.m. CET